Let's recap...
Recently, I published a few lists of Hacker/Infosec/Cybersec Discord servers.
Hacker_Discords
&
Hacker_Hiring_Discords
To compile these lists meant joining MANY Discord servers...really as many as I could.
~~ KNOW OF ONE? PLEASE ADD YOURS! ~~
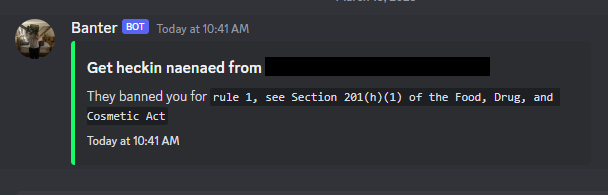
Upon joining the REDACTED server I was promptly sent the following message.
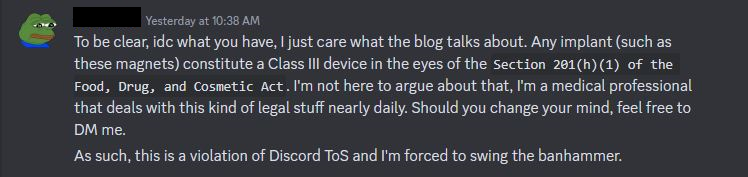
PEPE: Hi, I'm a mod from REDCATED server. I'm concerned that your profile description contains unapproved information regarding FDA Class 3 medical devices. Please remove the link or you will be banned.
Me:...
You're initial question likely being "What the heck is in your profile description?!". The answer being...a link to my blog. The one that you are reading. Hi :)
...and before I could respond the banhammer had already swung...
I don't think they were TRULY interested in me DM'ing them and us discussing the topic, ha. As an exercise though let's take a look at Section 201(h)(1) of the Food, Drug, and Cosmetic Act, FDA Class 3 medical devices, and this blog. The US Federal Food, Drug, and Cosmetic act is a set of laws passed in 1938 giving authority to the FDA to oversee the safety of food, drugs, medical devices, and cosmetics.
For reference I've uploaded a copy of the FDA publication to GitHub; Relevant pages below.
Extracting the important information we're told that a "device" is any instrument, apparatus, implement, machine, contrivance, IMPLANT, in vitro reagent, or other similar article which is:
- Recognized as a drug.
- Intended to diagnose, cure, or prevent disease - medical intervention.
- Intended to AFFECT THE STRUCTURE OR FUNCTION OF THE BODY, doesn't achieve purpose through chemical action, and isn't dependent upon being metabolized - medical intervention.
- The term "cosmetic" meaning intended to be rubbed, poured, INTRODUCED INTO, or otherwise applied to the body or any part part for cleansing, beautifying, attractiveness, or altering appearance.
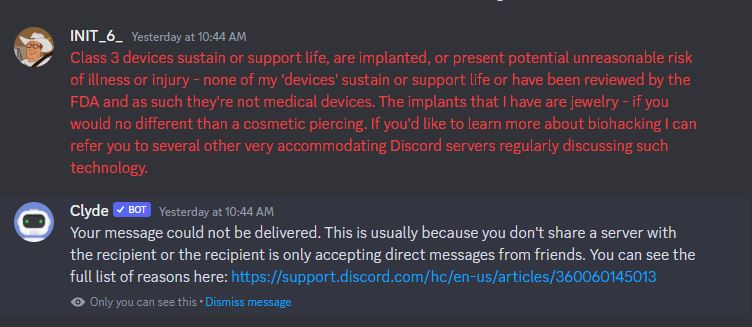
Note that the FDA does not approve cosmetics with the exception of color additives. Otherwise, there are three key elements at play. The word "implant", the phrase "affecting the structure or function of the body", and the cosmetic phrase "introduction into". As for the contents of my blog, sans the FDA approved VeriChip medical device, the closest match to these factors are implantable magnets. Magnets being an implant intended to give the user a degree of electromagnetic perception or the ability to lift ferromagnetic objects. However, none of the implants have any direct affect to preexisting body functions or change any existing structures. Current biohacking implants are no more than a piercing, i.e., body mod in the eyes of the FDA. Body mod with extra steps. Not that biohacking has ever advertised itself as 'FDA approved' to begin with ¯\_(ツ)_/¯
But wait you're saying, "How do we know for certain that biohacking is body mod?". Luckily for us the Association of Food and Drug Officials a.k.a also the FDA already answered that question. See GitHub for the full FDA Law and the Body Art Industry presentation.
Biohacking, in the context of transdemal implants, is unilaterally considered by the FDA to be a body art and as an extension a form of personal expression - not a medical device or consumer product. Nueralink requires an FDA approval because it's a consumer medical device - which they still have not received. Biohacking doesn't seek approvals - innovate first, questions second, safety third...or something like that. Getting banned from a Discord Server over a misunderstanding of biohacking and the FDA seems pretty silly but the potential for a REALLY bad time exists.
Want more info on the topic? Check out the book Bodies under Siege, the Dangerous Discord, or Augmentation Limitless.
[BIOHACK THE PLANET]





Comments
Post a Comment